Xizimed is a CRO specializing in registration of APIs, excipients and packaging materials and compliance consulting services, including Shenyang Xizi Biomedical Technology Service Co., Ltd. and its subsidiary Beijing Xizimed Consulting Inc. We are committed to providing domestic and foreign customers with the most professional and precise registration and compliance consulting services for APIs, pharmaceutical excipients and packaging materials, to build the most time-saving and labor-saving approaches of listing.
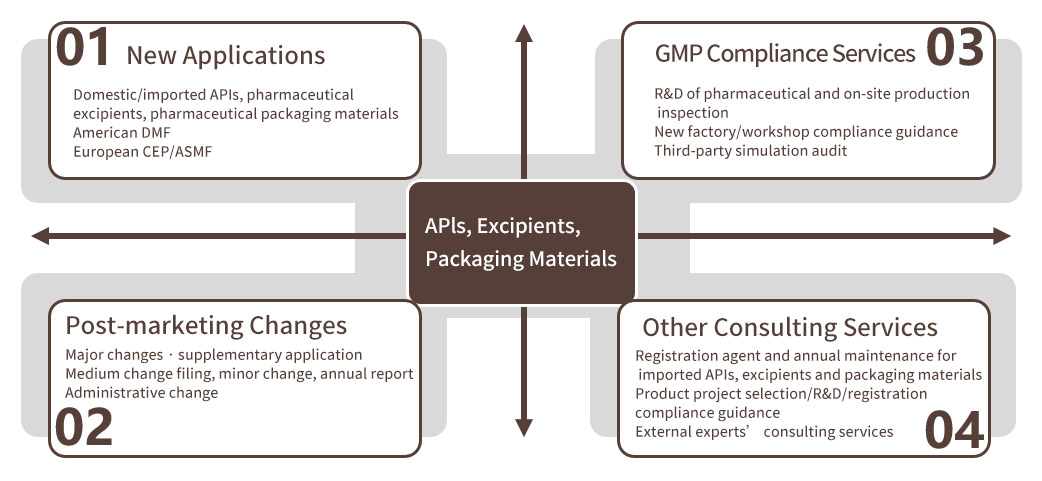
The main business of Xizimed includes:
→Import and export registration of APIs
Import and export registration of APIs is the core business of Xizimed, including import registration of produced overseas APIs to CDE, and export registration of domestic APIs to Europe and America (CEP/ASMF/DMF).
→Import and export registration of excipients and pharmaceutical packaging materials
It includes the import registration of DMF products produced overseas to CDE, the export registration of domestic products to Europe and America, and the relevant change and maintenance services of registered products.
→Drug R&D registration compliance consultation
Based on years of work experience of drug R&D, production and quality management, as well as a large number of practical cases of deep understanding and experience summary, Xizimed can provide domestic and foreign customers with new product project selection, auxiliary process development, guidance for the establishment of specifications and impurity research, assistance in formulating the optimal change strategy for marketed products and other technical consultation and compliance services.
Xizimed has rich experience and successful cases in the field of R&D and registration of APIs, pharmaceutical excipients and packaging materials. In addition to conventional chemical synthesis products, we also have registration experience in fermentation, extraction, multi-component/polymer excipients, complex packaging system, and even cell-related products, culture media, vaccine adjuvants, etc.
Xizimed has been deeply involved in the field of APIs, pharmaceutical excipients and packaging materials for many years, and is willing to become the trustworthy professional regulatory consultant for APIs, excipients and packaging materials for the global medical and health industry!









